John P. Hammond
Starna Scientific Limited, 52/54 Fowler Road, Hainault, Essex IG6 3UT, UK. E-mail: [email protected]
Regular readers of the Quality Matters column will be aware that the world of Quality Assurance is a constantly evolving environment. This evolution causes existing standards to be periodically reviewed, revised and utimately reissued. To those of us involved in this review process, these revisions inevitably raise questions during the process, and when the standard in question is internationally recognised, revision does promote some interesting discussions at times. However, in time, consensus is reached, the revised standard issued and then the process of informing the user-base of the “what, why, how” of compliance to the new standard begins.
Such a scenario was reported in this column earlier this year, where we discussed the modernisation of the spectroscopic General Chapters in the United States Pharmacopeia (USP). Clearly this is a major revison of the USP, and now the impact of these changes in being felt in the user community, and the obvious questions raised. Not surprisingly, this highly regulated environment now states that:
“Wherever possible in the procedures detailed as follows, certified reference materials (CRMs) are to be used in preference to laboratory-prepared solutions. These CRMs should be obtained from a recognized accredited source and include independently verified traceable value assignments with associated calculated uncertainty.”
So now, for the first time in these chapters, we see specific reference to CRMs, no pun intended.
Where ISO standards are involved that specifically require the use of reference materials (RMs), e.g. ISO/IEC 17025 etc., the ISO technical committee responsible for the “what, why, how” is ISO/REMCO and the work of this committee has often been descibed in this column.
Unsurprisingly, ISO/REMCO is also paralleling this evolutionary path, and the changes to this organisation’s work programme following its recent annual meeting in Pretoria, South Africa, are discussed below.
On 9–12 June 2015, the ISO Committee on Reference Materials (ISO/REMCO) held its annual meeting by invitation of the South African Bureau of Standards (SABS). In the convivial surroundings of their conference suite in Pretoria, there was a structured programme of plenary sessions and working group meetings, which also initiated many constructive deliberations and debates into the topics under review.
The focus of the annual meeting was centred upon two key areas. First, the stated revision in the work programme of ISO/REMCO, and second, the conversion of ISO Guide 34:2009 to ISO 17034, and the normative and advisory support guides to this new standard, which are being updated to match current requirements.
Evolution of “ISO/REMCO”
As a technical committee of ISO, ISO/REMCO was formed in 1975, principally to address the lack of guidance with respect to the production, use and certification of RMs. Unsurprisingly, the first Guide produced by this committee in 1981, ISO Guide 30, addressed the specific terminology associated with RMs. This process continued through the 1980s and 1990s, until the set of ISO Guides (31 to 35) was completed with the publication of ISO Guide 34—“General requirements for the competence of reference material producers” in 1996; and then “just like painting a major transport bridge”, the (revision) cycle began at the start. However, this first set of Guides were produced purely as guidance documents, aimed to provide non-mandatory technical assistance to RM users and producers.
In the intervening period, as discussed above, evolution of these Guides has resulted in the latest version of ISO Guide 34 published in 2009, effectively being used as standard by laboratory accreditation bodies, and therefore the long awaited conversion to a formal ISO standard (ISO 17034) has begun.
This conversion does have ramifications in respect to the normative references associated with ISO Guide 34/ISO 17034, in as far as ISO Guide 30 will now have a mandatory aspect when considered as a normative reference to ISO 17034. In addition, mandatory aspects listed as simple statements in, and essential to, the implementation of ISO 17034 will be expanded and discussed more fully in the new revisions of ISO Guide 31 and ISO Guide 35, and therefore, where appropriate, these guides will use “shall” if the explanatory text relates to an associated ISO 17034 clause.
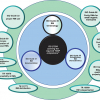
So now, with the conversion of ISO Guide 34 to ISO 17034, the ISO/REMCO “guides” have diverged into three areas:
- those listed as normative references, supporting ISO 17034, namely ISO Guide 30;
- those guides, that contain additional guidance on the essential (mandatory) requirements of ISO 17034, namely ISO Guide 31 and ISO Guide 35;
and
- those guides which follow the original scope of ISO/REMCO to simply provide additional (non-mandatory) technical guidance for a given process involving RMs; i.e. ISO Guide 33 and ISO Guide 80.
In addition ISO/REMCO has an established protocol for producing “TR” Technical Reports, via Ad-Hoc (AH) Working Groups, where the external request for guidance on a specific topic has been requested. This has resulted in the following four TRs, within the ISO/REMCO portfolio:
- TR 79: “Reference Materials for qualitative analysis—Examples of reference materials certified for nominal properties”;
- TR 10989: “Reference Materials—Guidance on, and keywords used for, RM categorization”;
- TR 11773: “Global distribution of reference materials”;
- TR 16476: “Reference materials—Current status of establishing and expressing metrological traceability of quantity values assigned to reference materials”.
Therefore, in order to again address these new requests for guidance is specific areas, ISO/REMCO has created two new AH groups, tasked with investigating and reporting the following:
- AHG4: “Development of guidance on characterization of pure materials”;
- AHG5: “Development of new approaches to the establishment and monitoring of Stability and Homogeneity”. The provisions in ISO Guide 35, by definition, can only provide examples of the many protocols available, and therefore there is value in reviewing current practice(s). In the longer term this might allow ISO Guide 35 to be simplified to a set of principles with detail in separate documents.
AHG4 and AHG5 have been requested to prepare reports on their review of corresponding existing documents, consultations with relevant committees and bodies, inside and outside ISO, as well as on proposals for the way forward, for discussion during the 39th REMCO meeting (June 2016). The explanation of the technical programme is shown graphically in Figure 1.
In addition to these new discussion items, as stated above, the current ISO/REMCO work programme covers the following Guides and Technical Reports in support of ISO Guide 34:2009/ISO 17034.

ISO Guide 30
The current revised version contains definitions for “candidate reference material” (material, intended for future use as an RM), “primary measurement standard” and “secondary measurement standard”. It also defines “indicative value” and “information value” as values associated with a CRM. ISO Guide 30was published in October 2014.
ISO Guide 31
ISO/REMCO has ISO Guide 31, which advises on the contents of certificates, labels and accompanying documentation for RMs. The new guide contains requirements not only for RM certificates, but also for product information sheets (i.e. information sent with non-certified RMs). Naturally, requirements for certificates are more comprehensive. Mandatory information of RM certificates are besides the certified values and their uncertainty, intended use, minimum sample intake, validity period of the certificate (in the current version only needed if degradation is deemed possible), storage information, instruction for handling and some administrative information. RM certificates require a clear specification of the measurand and the reference to which the values are traceable to, and where the value assignment is method specific, the methodology used, e.g. for particle characterisation, this means that the methods for which the certified value is valid need to be clearly stated on the certificate, if the material is such, that different methods will give different values. The current revision of this document has been accepted, with minor changes discussed at the meeting, and will therefore be published later in 2015.
ISO Guide 35
ISO Guide 35, as a key document supporting the accreditation of RM producers, is now extensively being revised. As part of this revision process the guide has been renamed “Reference Materials—Guidance for the assessment of homogeneity and stability, as well as the characterisation of the material”. The current draft revision was reviewed at the meeting, where key areas of contention were discussed. The decision was taken that following a teleconference in July 2015 to review an updated draft, the proposal is to submit the document to the ISO ballot process, with the intention to discuss the result of this action the next meeting in June 2016.
As discussed above and shown in Figure 1, the recent non-mandatory guidance documents recently completed by ISO/REMCO are detailed below.
ISO Guide 33
ISO Guide 33:2015, was published on 1 February 2015. ISO Guide 33:2015 describes good practice in using RMs, and CRMs in particular, in measurement processes. These uses include the assessment of precision and trueness of measurement methods, quality control, assigning values to materials, calibration and the establishment of conventional scales. This Guide also relates key characteristics of various types of RMs to the different applications. The publication of ISO Guide 33:2015 automatically withdraws ISO Guide 32:1997 (Calibration in analytical chemistry and use of certified reference materials) as its content has been relocated into the new ISO Guide 33.
ISO Guide 80
Published in April 2014, ISO Guide 80 outlines the essential characteristics of RMs for metrological quality control (QCMs), and describes the processes by which they can be prepared by competent staff within the facility in which they will be used (i.e. where instability due to transportation conditions is avoided). The layout and structure of this Guide provides general information on the preparation of QCMs in the main chapters, with specific case studies covering a range of sectors in the annexes. The case studies are not complete “process manuals” but are included to highlight some of the key considerations when preparing QCMs. The case studies vary in complexity and detail, including sector specific terminology, but provide a range of information for laboratory staff to draw from.
TR 16476: Reporting Metrological Traceability
A revised “final” draft DTR 16476 (which had been balloted) was reviewed at the meeting, and once a few minor changes have been made, this report will be submitted to ISO for publication this year (2015).
Conversion of ISO Guide 34 into ISO 17034
Within ISO, a joint Working Group between ISO/REMCO and ISO/CASCO (the technical committee responsible for issues relating to conformity assessment) has been established, for the conversion of ISO Guide 34 into ISO standard 17034.
The first meeting in early December 2014 produced a ‘”Committee Draft” (CD) standard, which was duly voted on, and the extensive comments received reviewed at this meeting.
It was expected that this conversion would follow a similar route to that of the ISO Guide 43 for Proficiency Testing to ISO standard 17043. However, as in any evolutionary process, this transition has been complicated by a required updating of the general structural guidance for all the “17000 series” standards under the control of ISO/CASCO, to be implemented here, and in the updating of ISO/IEC 17025, currently also under revision.
The next meeting of ISO/CASCO/JWG 43 took place on 7–9 July 2015 at ISO in Geneva, Switzerland, and at which CASCO/JWG43 decided to move its 17034 project “General requirements for the competence of reference material producers” to an ISO ballot from October 2015 to December 2015.
So now with this revision cycle of the “3x series” guides coming to an end, evolution has taken ISO/REMCO down a long awaited path, namely the conversion of ISO Guide 34 to ISO 17034, and in a similar cycle manner to this article;
“…consensus is reached, the revised standard issued, and then the process of informing the user-base the ‘what, why, how’ of compliance to the new standard begins.”